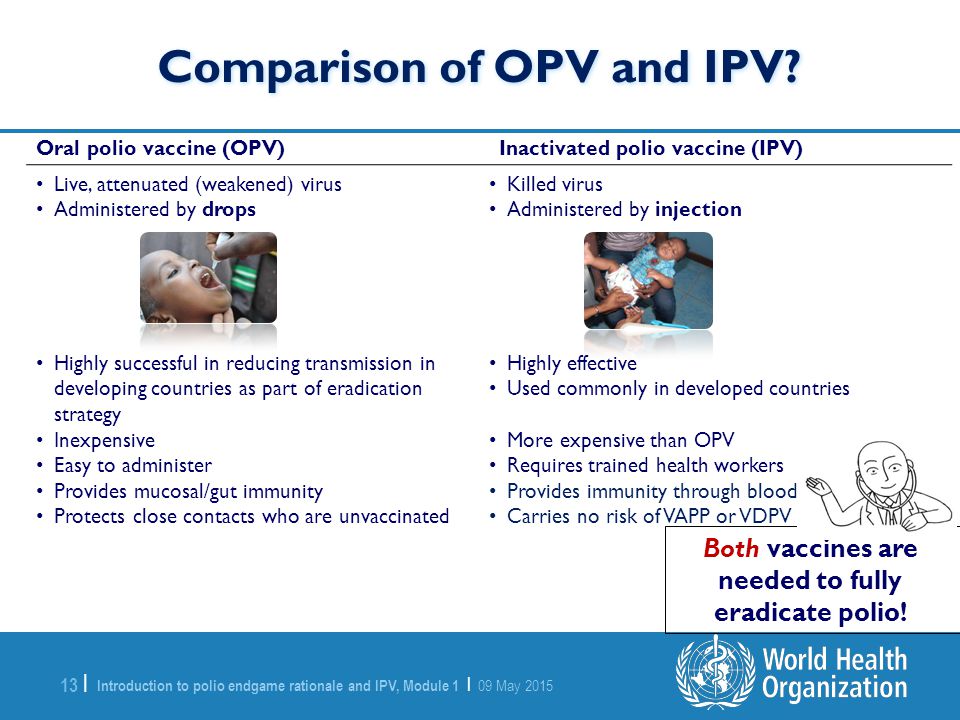
Potential Future Use, Costs, and Value of Poliovirus Vaccines - Thompson - 2021 - Risk Analysis - Wiley Online Library
Combined Immunization of Infants with Oral and Inactivated Poliovirus Vaccines: Results of a Randomized Trial in The Gambia, Oma

Economics of polio vaccination in the post-eradication era: should OPV-using countries adopt IPV? | Semantic Scholar

Economics of polio vaccination in the post-eradication era: should OPV-using countries adopt IPV? | Semantic Scholar

Safety and immunogenicity of inactivated poliovirus vaccine schedules for the post-eradication era: a randomised open-label, multicentre, phase 3, non-inferiority trial - The Lancet Infectious Diseases
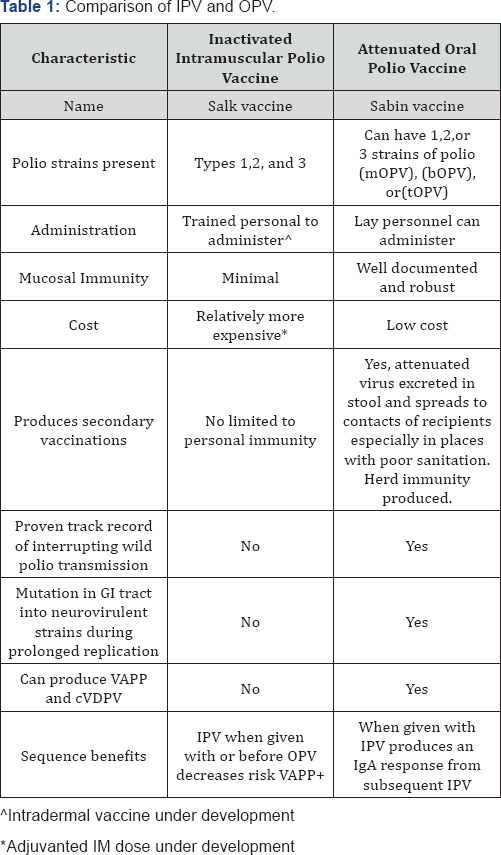
![PDF] Transmissibility and persistence of oral polio vaccine viruses: implications for the global poliomyelitis eradication initiative. | Semantic Scholar PDF] Transmissibility and persistence of oral polio vaccine viruses: implications for the global poliomyelitis eradication initiative. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/27701a7fa20c5b25ea4c98e45f9c39b07769d457/5-Table1-1.png)
PDF] Transmissibility and persistence of oral polio vaccine viruses: implications for the global poliomyelitis eradication initiative. | Semantic Scholar